The virus that causes COVID-19, like all viruses, isn’t a living organism; it isn’t even a cell. It’s a nanoparticle composed of 3 building blocks: a genetic control center (in this case, RNA) wrapped in a fatty lipid membrane (the viral protective coat) covered with protein spikes (the key to a host cell’s door). The COVID-19 virus, otherwise known as SARS-CoV-2, is a mutated version of coronavirus that is more virulent than any coronaviruses we have seen in the past.
Let’s start with a little trivia because it can get confusing: Viruses and diseases often have different names. For example, HIV is the name of the virus that causes the disease we call AIDS. Similarly, SARS-CoV-2 is the official name of the COVID-19 virus that causes the disease, we call COVID-19. As far as wildcards go, SARS-CoV-2 (aka COVID-19 virus), was dealt aces high because it is covered in protein spikes that bind angiotensin-converting enzyme-2 (ACE-2) proteins.1 To our misfortune, ACE-2 proteins are ubiquitously found covering cells all over our respiratory and gastrointestinal tracts, as well as in our kidneys and heart. The spikes serve as the virus’ key, to make contact with ACE-2 proteins on the surface of our cells. Next, it uses an enzyme found in the host cell – called furin – to cleave its spikes in half, opening the lock, allowing the virus to sneak into your cell undetected. Once inside, the virus quietly hijacks your cell to replicate more viral units.
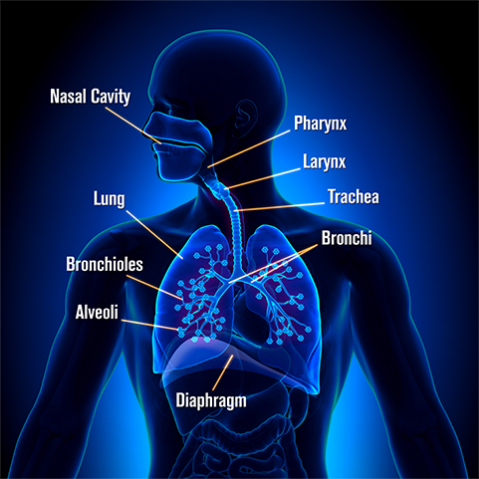
The viral load and how long it takes for your immune system to figure out something isn’t right, influences how sick you will get from infection by COVID-19.
Our immune system has never seen anything like COVID-19 before, which is why this virus is so good at slipping in under the radar. In the beginning, the virus silently replicates before your immune system knows something is wrong. Once the viral load gets too large, the host cell bursts releasing millions of viral minions to propagate the infectious cycle. The viral load and how long it takes for your immune system to figure out something isn’t right, influences how sick you will get from infection by COVID-19.
In an ideal world, your immune system can contain this virus up north in your nose and sinuses, which would keep symptoms mild, limited to low-grade fevers, fatigue, and/or a decreased sense of smell. Nasal congestion, a runny nose, or progression down to the pharynx and throat, causing a sore throat, can also occur, although this was reported in as few as 4-5% of COVID-19 positive patients.2 More commonly, the virus travels south to irritate our larynx (voicebox), causing a cough in as many as 82% of patients.2 This is when we grow more concerned, as further progression down the trachea (windpipe) to our bronchioles and delicate alveolar sacs in our lungs can lead to a more complicated course of this disease. Shortness of breath, a deeper cough and chest tightness indicate inflammation of the bronchial tree and patients are commonly admitted for treatment at this point.
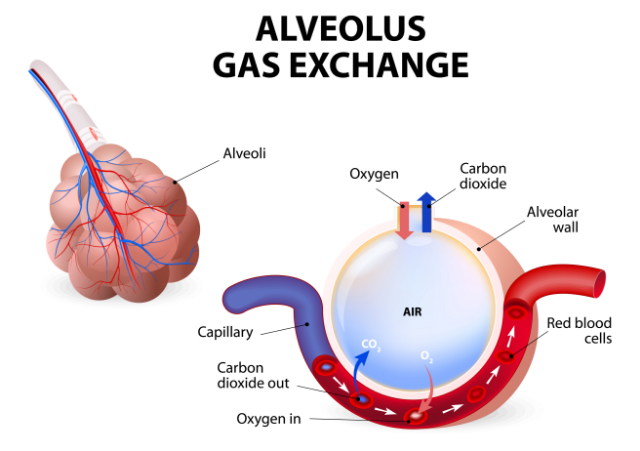
Obviously, the exchange of air in our delicate lung alveolar sacs is a critical life function. These alveoli are wrapped in a nest of blood vessels, arteries (red) and veins (blue). Alveoli expand as they take in the fresh oxygen we breathe and infuse that into our arteries to pump out oxygen to every cell in our body. In exchange, they extract carbon dioxide waste from our veins to help rid our system of it during exhalation.
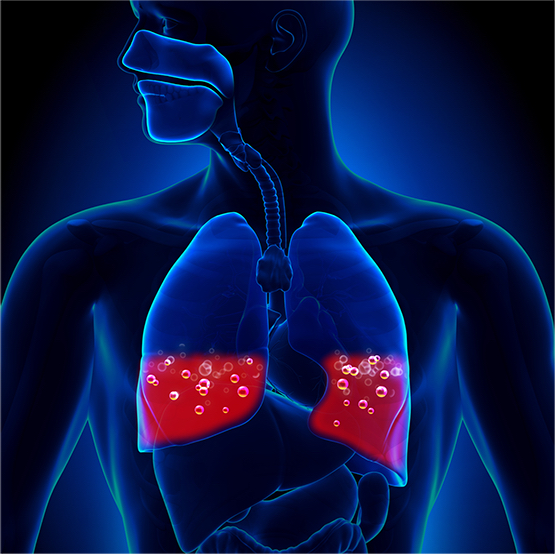
If the virus makes it down to the ACE-2 receptors in your alveoli, it can cause pneumonia, or worse – lead to the dreaded Acute Respiratory Distress Syndrome (ARDS). Pneumonia is caused by your alveolar sacs becoming so inflamed, fluid rushes in. As more of your respiratory cells die, the sacs become full of fluid, pus and debris, forming “consolidations” – areas of your lung where gas exchange becomes difficult or no longer possible. At this point, patients usually have high fever, chills, breathing difficulties, and a deep cough, often producing a yellow, greenish or bloody sputum. To make matters worse, other bacteria can enter the body while your immune system is down and cause “bacterial superinfections”, further challenging a patient’s recovery.
Once COVID-19 reaches your lungs, the immune system of certain individuals may go into hyper-drive, signaling an army of troops to help ward off further invasion. When the viral load is too great, or a person’s immune system is over-reactive, a frenzy of immune cells signals a host of molecules to annihilate the viral invasion. This seems like a prime situation, but in the wake of this frenzy we call cytokine storm, the immune system may try to kill all our infected cells. When this happens all at once, the dreaded Acute Respiratory Distress Syndrome (ARDS) is the result. As this condition arises from lung damage, patients tire out from breathing, go into respiratory failure and require a mechanical ventilator to keep them alive. As the condition progresses, the lung damage becomes so great, the lubricant in our alveoli called surfactant dries up, and there are not enough functional alveoli to exchange oxygen at all, ventilator or not.
When our lungs, the organ where oxygen exchange occurs dies, sadly, there is just no hope. As low blood oxygen levels (hypoxia) sets in, carbon dioxide levels build up (hypercarbia), causing our own blood to drop to a dangerous pH and signaling our system is in a state of “shock.” An overwhelming cascade of inflammatory chemicals are released in response, over time hastening multi-organ system damage. With no choice but to allow this poisonous acidic blood to circulate and bathe our organs, a domino effect ensues as each of our organs, primarily our kidneys and heart are overworked to try to compensate.
Which came first, “the chicken or the egg?” is still under investigation. Depending on a person’s underlying conditions, inflammation of any organ might be at risk with COVID-19 infection. To make matters worse, we remain uncertain whether the virus itself may hasten things by targeting ACE-2 proteins in distant organs such as the heart directly, or whether these complications are an indirect result of cytokine storm, and the body’s overwhelming immune response. Regardless of the cause, as multi-organ system damage progresses, there comes a point of no return and patients may die from any combination of pulmonary, renal, liver and cardiac damage, even while mechanically ventilated.
While, there is still much we do not know about the COVID-19 virus, each week we learn more and more about this disease. As of April 8, 2020, the Centers for Disease Control (CDC) reports the hospitalization rate for COVID-19 is 4.6 per 100,000 population, 74.5% of hospitalized patients were aged 50 or greater, and almost 90% of hospitalized patients have some type of underlying condition.3
Knowing these facts, and understanding how COVID-19 kills, helps us make sense of the horrifying data we are faced with on the frontlines. Strategies that involve competitive inhibition, like recombinant human ACE-2 protein4 and monoclonal blocking antibodies to the viral ACE-2 protein spike5 are just a few of the many strategies being developed in our race for a cure. For now, prevention is our only armor and we must use it to buy time until science catches up.
- 1. Tai W, He L, Zhang X, et al. Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell Mol Immunol (2020). https://doi.org/10.1038/s41423-020-0400-4
- 2. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. The Lancet (2020).https://doi.org/10.1016/S0140-6736(20)30211-7
- 3. Garg S, Kim L, Whitaker M, et al. Hospitalization Rates and Characteristics of Patients Hospitalized with Laboratory-Confirmed Coronavirus Disease 2019 -COVID_NET, 14 states, March 1-30, 2020. MMWR. ePub: 8 April 2020. http://dx.doi.org/10.15585/mmwr.mm6915e3external icon
- 4. Guo J, Huang Z, Lin L, et al. Coronavirus Disease 2019 (COVID‐19) and Cardiovascular Disease: A Viewpoint on the Potential Influence of Angiotensin‐Converting Enzyme Inhibitors/Angiotensin Receptor Blockers on Onset and Severity of Severe Acute Respiratory Syndrome Coronavirus 2 Infection. JAHA (Apr 2020).https://doi.org/10.1161/JAHA.120.016219
- 5. Shibo J, Hillyer C, Du L. Neutralizing Antibodies against SARS-CoV-2 and Other Human Coronaviruses. Trends in Immunology (Apr 2020) https://doi.org/10.1016/j.it.2020.03.007